Titanium emerges as a vanadium alternative for redox flow batteries
A Japanese–Chinese team developed a titanium molten salt redox-flow battery using abundant titanium ions and molten salt electrolytes to enable high-voltage, fast, and stable grid-scale energy storage. The system demonstrates high efficiency of over 97%, strong cycling stability, and improved cost and scalability compared to vanadium-based batteries, with further optimization underway.
April 8, 2026 Emiliano Bellini
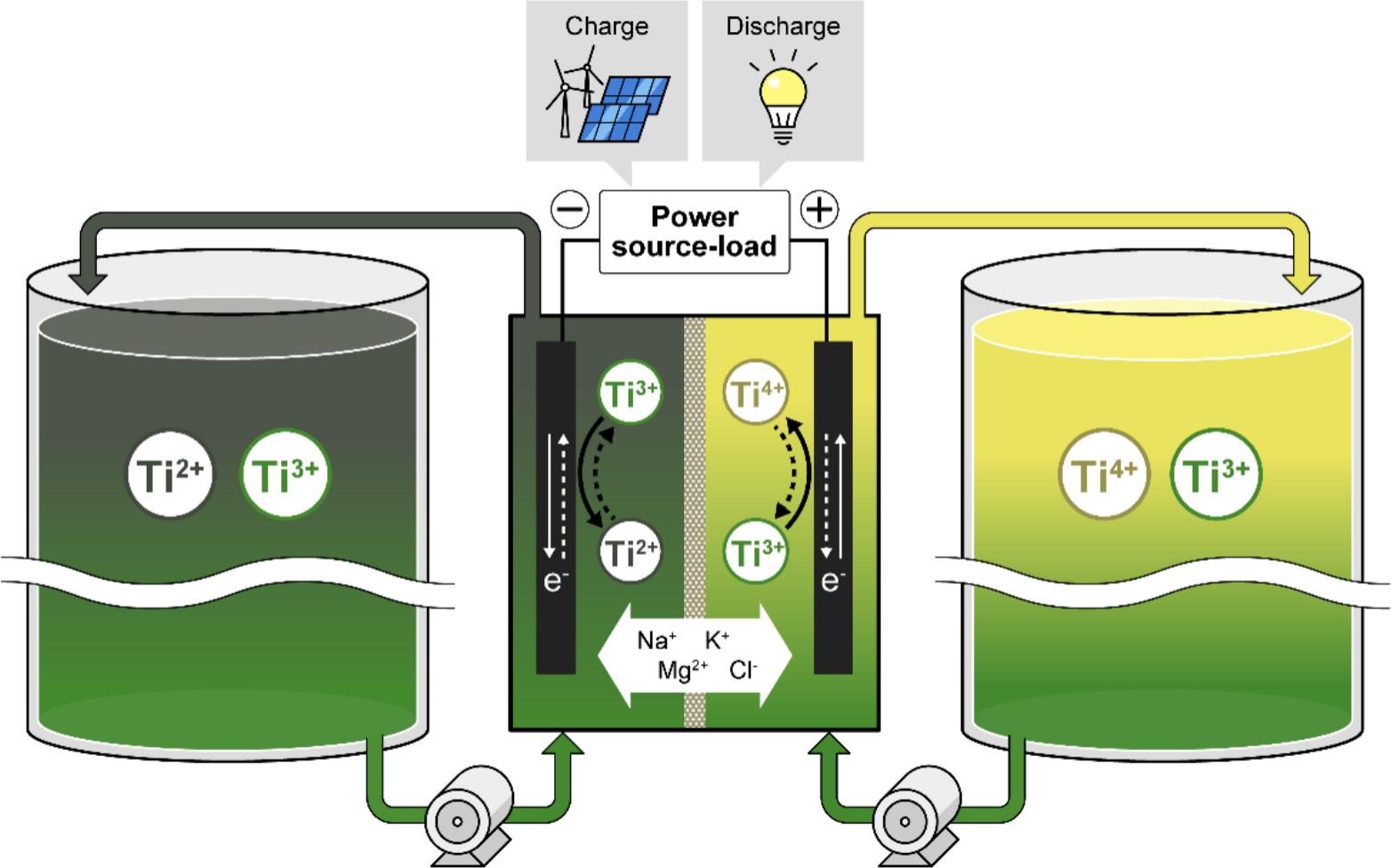
Schematic diagram of the developed titanium molten salt redox-flow battery
Image: Beijing University of Technology, Electrochemistry Communication, CC BY 4.0
A Japanese-Chinese research team has developed a titanium molten salt redox-flow battery (TMSRB) that uses titanium ions as the redox-active material and molten salt as electrolyte.
Intended for use in grid-scale energy storage, the TMSRB is designed to provide higher charge-discharge current density compared to conventional vanadium redox flow battery (VRFB) designs.
The scientists explained that, compared to vanadium, titanium is a far more abundant element, addressing supply and cost limitations. “Titanium is the seventh-most abundant metal in the earth's crust, with a crustal element abundance of 0.56%, 35 times that of vanadium. Thus, there is no concern about the sustainable supply of redox-active materials in TMSRB,” they stressed.
The system operates using titanium ions in multiple oxidation states, with the Ti⁴⁺/Ti³⁺ redox couple at the cathode and the Ti³⁺/Ti²⁺ redox couple at the anode, enabling reversible redox reactions. It also employs molten salt electrolytes, such as lithium chloride–potassium chloride (LiCl–KCl) and sodium chloride–magnesium chloride–potassium chloride (NaCl–MgCl₂–KCl), which the scientists said offer a wide electrochemical stability window and high ionic conductivity, while supporting efficient and high-voltage operation, rapid charge-discharge rates, and stable cycling at temperatures between 300–450 C.
.....................................






